SARS-CoV-2 Mu Variant PCR assays
The Mu variant (lineage B.1.621 or VUI-21JUL-1) of SARS-CoV-2 is considered as Variant of Interest (VoI) by the WHO since August 30.2021 because it carries mutations that indicate a risk of resistance to vaccines.
The Mu variant is mainly found in Columbia and South America but has been found in 41 countries so far, including most European countries , the USA and Canada.
We offer the following assays for the detection of the Mu Variant
1) Screening Assay -simultaneous SARS-Detection and VoC Determination of Delta and Mu
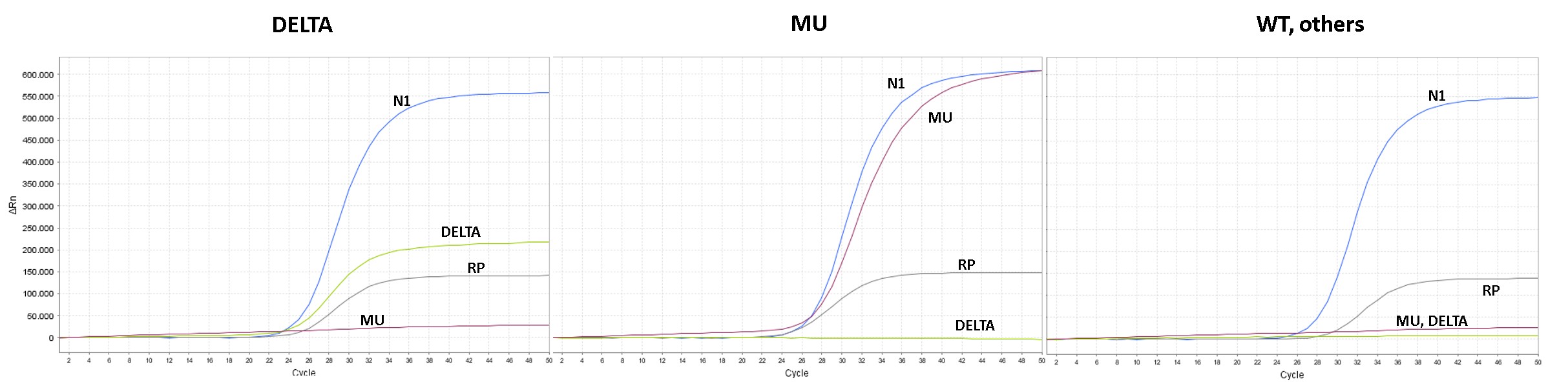
Detects SARS-CoV-2 via the N1 gene and determines the Mu and the Delta VoC in a single PCR
Since both Variants together represent more than 90% in some countries, this assay directly detects the VoCs of most positive samples in a single PCR. For non-Delta and non-Mu variants, our “all VOC” assay can be performed as follow-up PCR
Above: PCR assay for SARS-CoV-2 screening and simultenous determination of Delta and Mu VoC in a single assay with a dual target approach. The assay also includes a sampling or optionally internal control for highest relaiabilty.
Cat. Nr. 031522-DELTA-MU; 96-10.000 reactions
2) Mu Variant detection assay
- Detects the Mu Variant and non-Mu Variants in a dual-plex assay
3) Mu Variant and Alpha, Delta and Gamma -Assay (ADGM-Assay)
- Specifically determines Alpha, Delta and Gamma and Mu strains ina quad-plex assay
Cat. Nr. 031522-ADGM 96-10.000 reaction
All assays use FAM, HEX (or optionally JOE) ROX and Cy5for available on most qPCR machines.
Our assays have been tested on Abi7500, AbiStepOne, Qiagen Rotorgene, MIC, BioRad CFX.
For all other SARS-CoV-2 related product including PCR tests for specific mutations to determine the Lambda, C.1.2 as well as other VoCs and VoIs please click HERE.
All assays are currently for Research Use Only, certification process is ongoing and our kits are already in use after internal testing of certified laboratories.
Contact
For more information or to purchase our kits or services, please write us an e-mail (info@genxpro.de), or call us at (+49 69) 95739710 ).