VoXcreen qPCR assays
Our PCR tests robustly target distinct deletions in the genomes of all Variants of Concern.
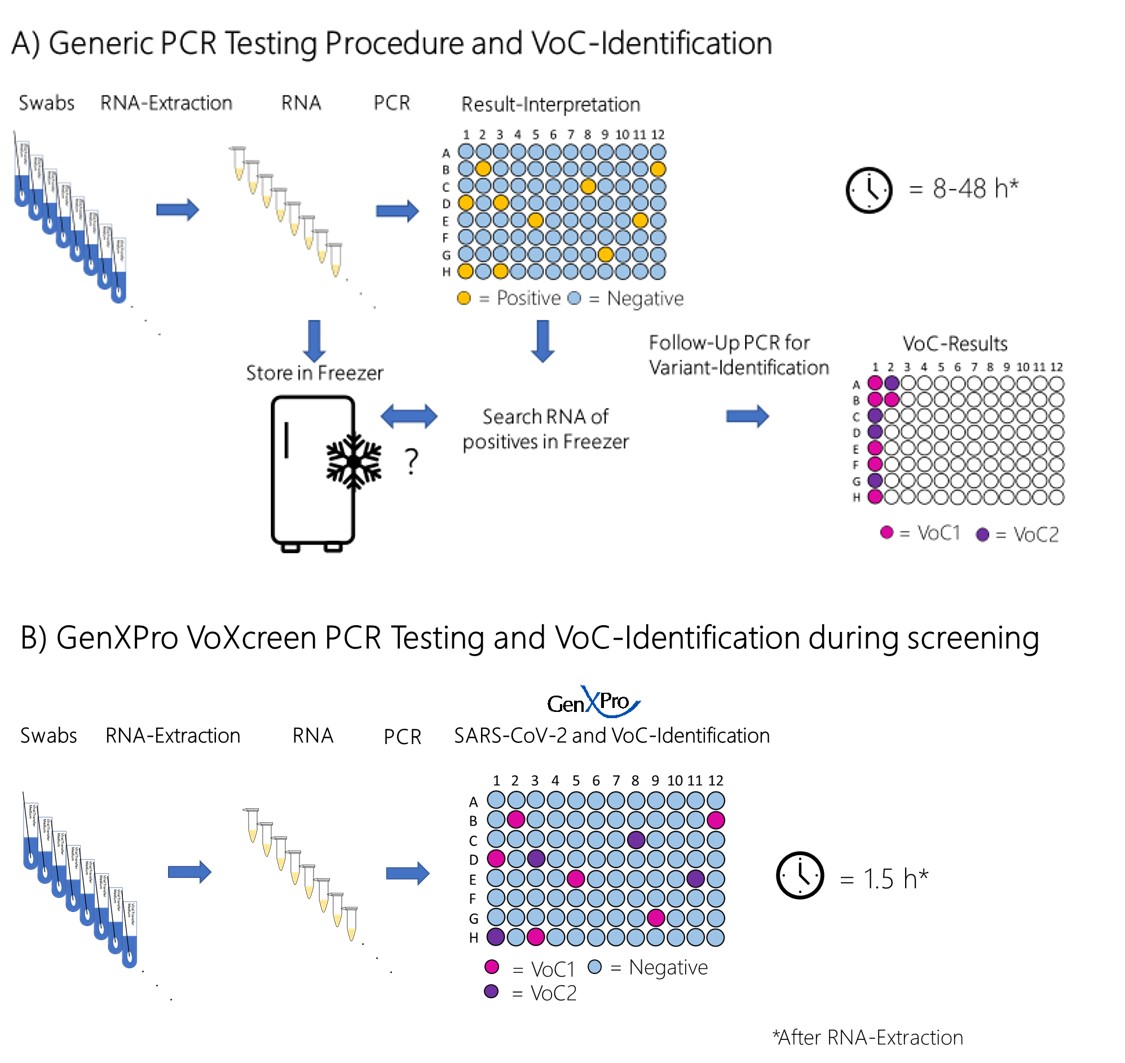
Our VoXcreen assay is designed to identify during the SARS-CoV-2 screening PCR the three most prevalent currently circulating variants, responsible for >99% of the infections.
Since June 2022 our latest test distinguishes between the Omicron subvariants BA.2 (21L), BA.4 (22A) and BA.5. (22B). We also offer a specific assay for BA.2.12.1 (22C). These strains represent nearly 100% of all circulating strains in the in most countries of the world.
Our assay detects universal sites that are present in all SARS-CoV-2 variants, using a WHO-recommended assay design, in addition to the VOC-specific assays. Therefore, all samples are identified with the recommended dual-target strategy. The assay also contains an internal isolation control (RP).
While current tests need at least two separate PCR reactions and often also genome sequencing for the specific identification of VoCs, our test identifies the variants reliably in a single PCR reaction. This saves time and liberates PCR-capacities that are critically needed.
Our VoXcreen BA.2-4-5 PCR assay was recently tested by the virology department of the University of Marburg and considered as very useful for the fast detection of SARS-CoV-2 variants.
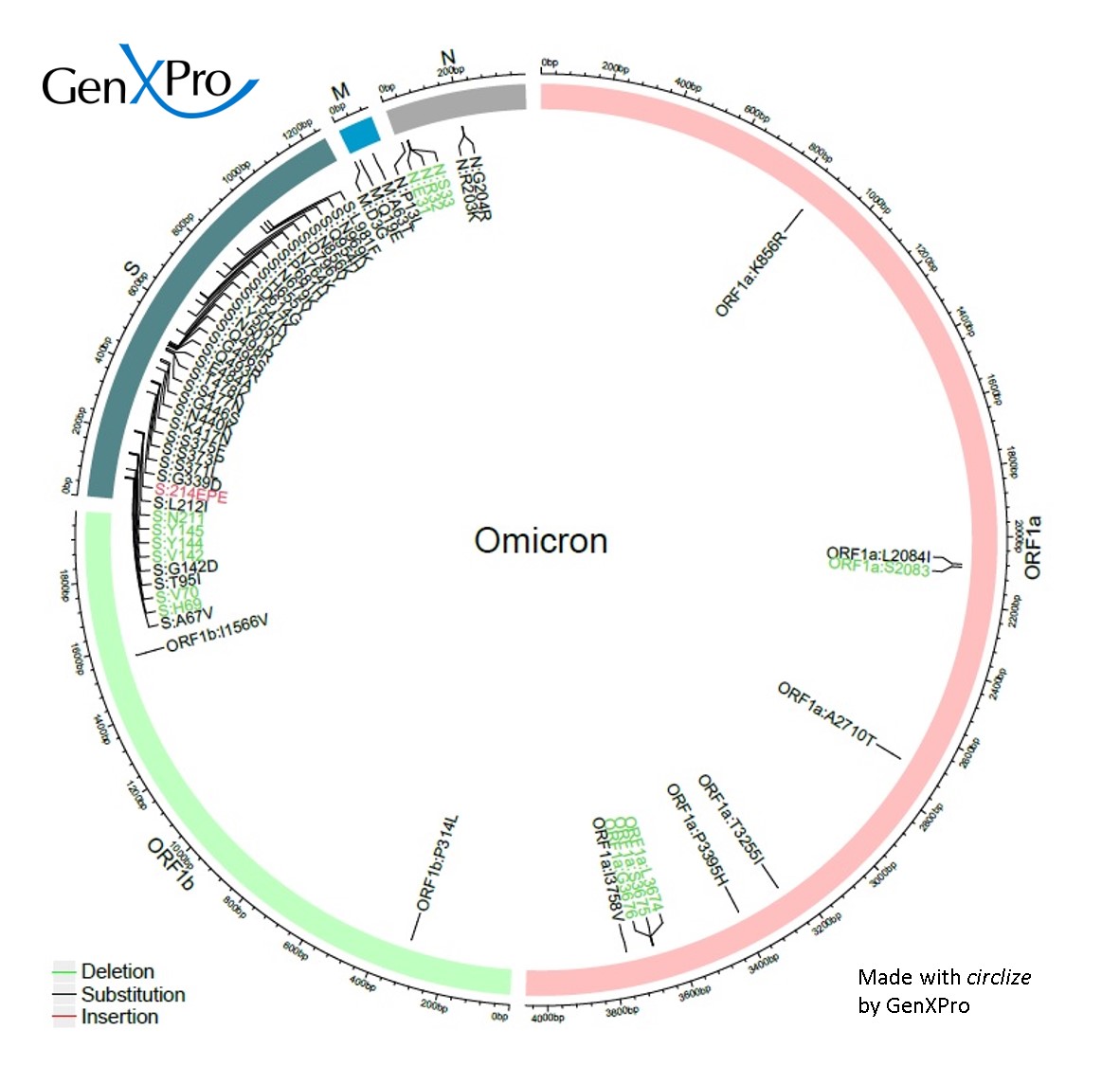
About the Omicron-VoC
The Omicron variant (B.1.1.529 is the latest Variant of Concern (VoC) of SARS-CoV-2, declared the 26th of November by the WHO. It was first identified in South Africa and was considered as VoC because of several local, steeply increasing number of infections. Moreover, it outnumbered the already highly infectious Delta variant in the concerned areas and therefore was quickly regarded as VoC by the WHO. The variant has is now the dominant variant in all countries. Currently, sub-variants of B.1.1.529 have already replaced the original strain, today (June 2022) BA.2, BA.5, BA.4 and BA.1.12.1 together comprise nearly 100% of all circulating strains wolrd-wide (https://covariants.org)
For orders- new kits in town
Cat. Nr. VXI-BQ11; 96-10.000 reactions (For VoXcreen BQ.1.1 identification+ internal ctrl.)*
Cat. Nr. VXI-BA461; 96-10.000 reactions (For VoXcreen BA.4.6.1 identification+ internal ctrl.)*
Cat. Nr. VXI-BF7; 96-10.000 reactions (For VoXcreen BF.7 identification+ internal ctrl.)*
*based on the identification of the mutations S:R346T; S:N460K and / or S:K444T
Cat. Nr. VXC-NDORP; 96-10.000 reactions (For VoXcreen N1, Delta and Omicron + internal ctrl.) – detects all Omicron variants.
Cat. Nr. VXC-NB2-4-5RP; 96-10.000 reactions (For VoXcreen N1, Omicron BA.2, BA.4 and BA.5 + internal ctrl.)
Cat. Nr. VXI-ADORP; 96-10.000 reactions (For VoXcreen Alpha, Delta and Omicron detection + internal ctrl.)
Cat. Nr. VXI-ORP; 96-10.000 reactions (For VoXcreen Omicron detection + internal ctrl.)- detects all Omicron variants.
Contact
For more information or to purchase our kits or services, please write us an e-mail (info@genxpro.de), or call us at +49 69 95739710.
For more SARS-CoV-2 / Covid PCR tests and sequencing solutions please click here.
(Click here for Page in German Language)
From tissue to results- full service or kits for all your samples

MACE kit
cfDNA-Seq Kit
Gel Free smallRNASeq kit
TrueQuant (UMI) Adaptor kits for up to 386 samples
16s-RNA Seq Kit for Microbiomes


